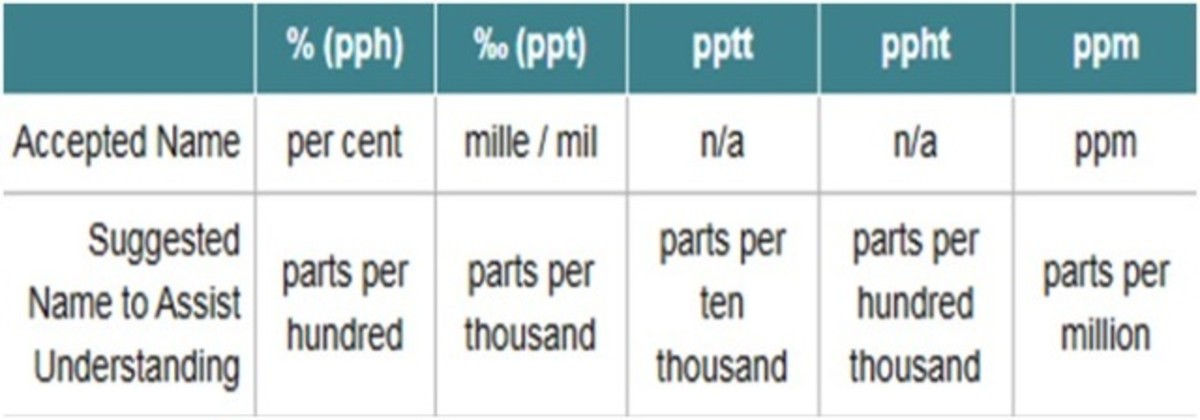
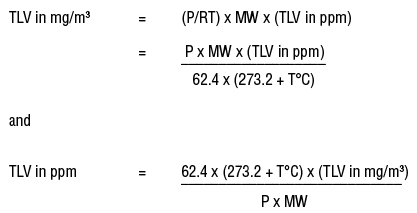
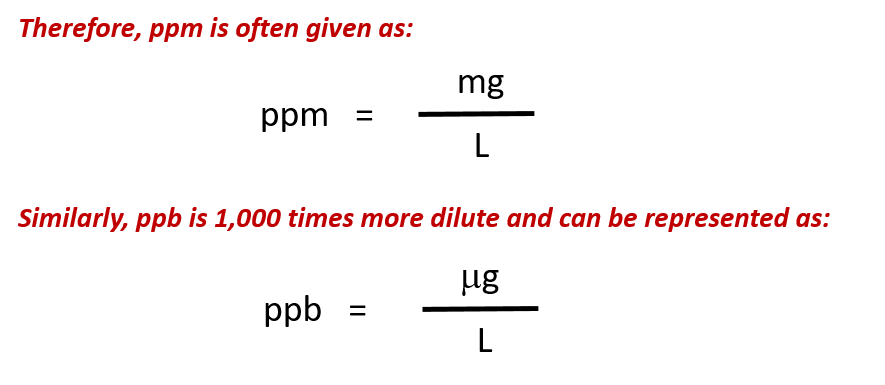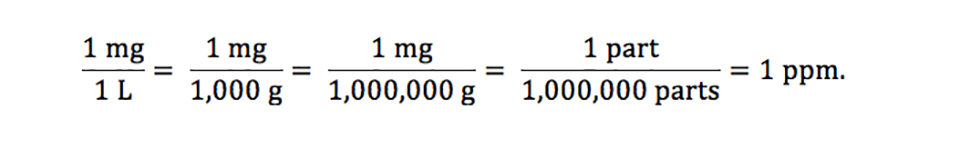Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download

Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download

Suppose a pure Si crystal has 5 * 10^28 atoms/m^3 at room temperature. It is doped by 1 ppm concentration of pentavalent As. Calculate the number of electrons and holes. - Sahay Sir